October is Breast Cancer Awareness Month, as many of you know, and we’ve been busy the past few...


October is Breast Cancer Awareness Month, as many of you know, and we’ve been busy the past few...

We are excited to announce that our AIROS Medical team will be participating in three conferences over the...
Listen to our CEO, Gerald Makoid, discuss the AIROS Medical product line and treatment of lymphatic disorders on...

We recently held a fun family day event with Lisa’s Army, our non-profit community partner!

We are thrilled to announce our partnership with Fist Assist Devices, LLC, creators of the FDA-cleared Fist Assist...

Our team will be in Phoenix, Arizona from July 12-14 to present our product line at Medtrade West...
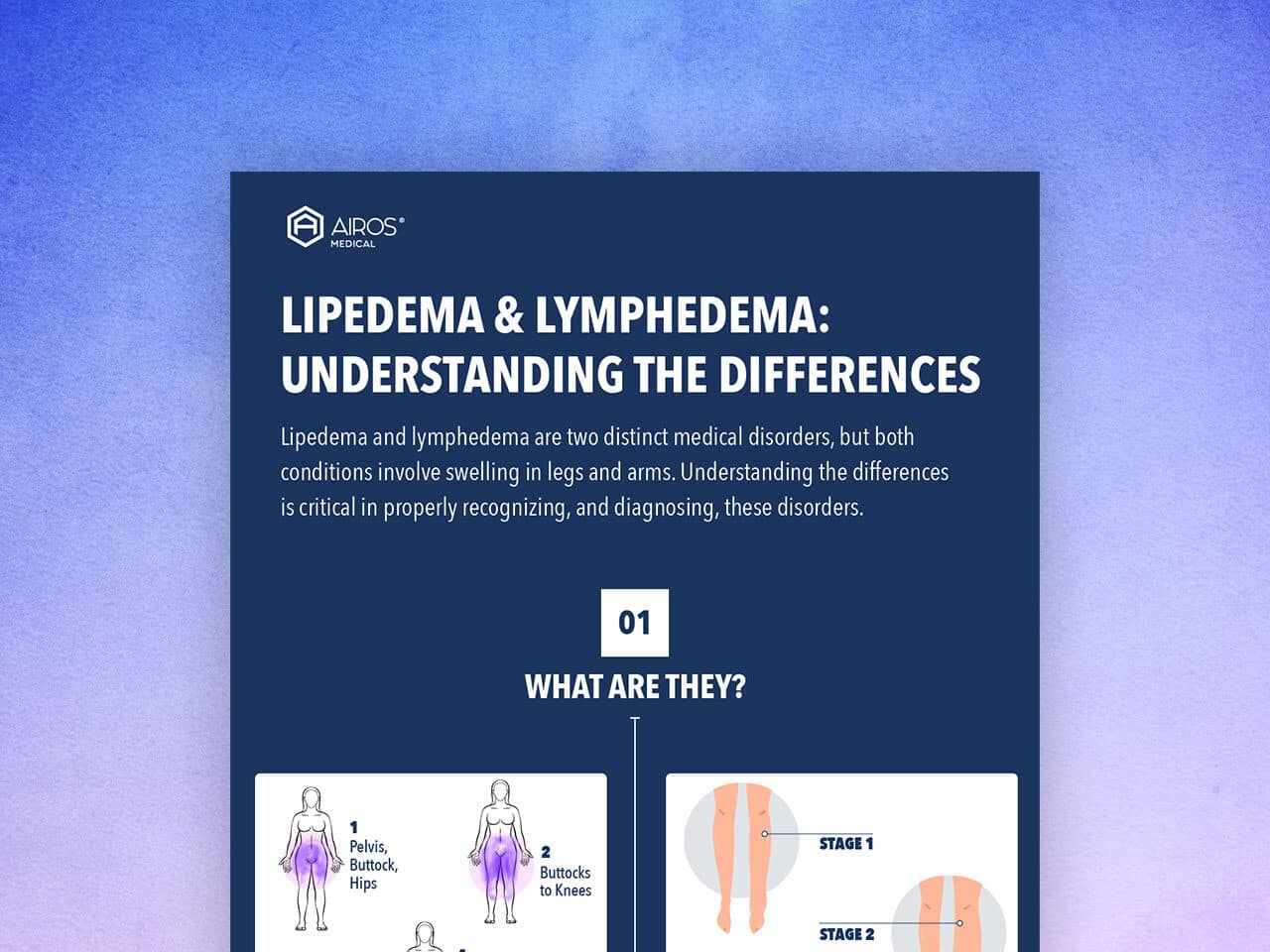
June is Lipedema Awareness Month. We are helping raise awareness about this misunderstood and misdiagnosed condition, as well...

In partnership with the Symposium on Advanced Wound Care (SAWC), AIROS is proud to support, sponsor, and co-present...

We are proud to announce our partnership with Lisa’s Army, a 501(c)(3) non-profit organization that provides comfort and...

We are proud to announce our 2021 Premier Sponsorship of the Lymphology Association of North America® (LANA)! LANA...

March is Lymphedema Awareness Month, during which the clinical, patient, and advocacy communities, as well as the private...

We had a successful showing at The Southern Association for Vascular Surgery’s (SAVS) 45th Annual Meeting in Scottsdale...